|
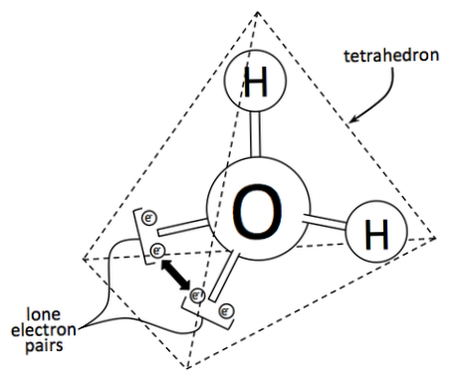
December is a time of skiing and sleigh rides in much of the northern hemisphere. Not so much in coastal California. While iconic snowflakes rarely fall on my garden, your situation may be something else entirely. But, being from New England, I’ve seen my share. It wasn’t until very recently that I learned just how amazing each snow crystal really is. Contrary to popular sentiment, bits of snow are not flakes. They are crystals. How these crystals form is mind-boggling. What it does to your garden may surprise you, too. But let’s start with a water molecule. Water chemistry Most of us are familiar with H2O. That moniker tells us that each molecule of water is made up of two hydrogen atoms connected to one oxygen atom. It ends up that all those whirling electrons are pulled closer to the oxygen atom. This gives oxygen a slightly negative charge, leaving hydrogen atoms with a slightly positive charge. [Don’t run away! This will all make sense in just a moment.] For snow crystals to form, water vapor, liquid water, and ice must all be present. As gazillions of water molecules float around in a cloud, their charges attract and repel each other like magnets. The negative side of a water molecule is pulled toward the positive side of other water molecules. [This is why water has surface tension, too.] When snow crystals form, five more water molecules, in liquid form, collect on this negative side and end up creating a 4-sided pyramid, or tetrahedron. As temperatures drop, this water turns to ice, suddenly shifting into the iconic 6-sided shape that decorates Christmas cards around the world. Up to this point, most snow crystals are identical. It’s when countless combinations of trajectory, humidity, and temperature changes are factored in that each snow crystal becomes unique. [Did you know that snow crystals can form triangles, diamonds, and pillars? I didn’t either.] Snow in the garden If you wake one winter morning to see your garden blanketed in snow, don’t panic. While freezing temperatures can kill tender annuals, making a mess of leaves, a thick covering of snow protects plant roots, earthworms, and important soil microorganisms. Those chilling hours translate into bigger fruit and nut crops next year, too! But how can snow crystals protect plants from freezing? It doesn’t make sense! As temperatures drop, the water contained in plants expands as it freezes. This causes frost cracks in stems and blackened leaves. Snow-covered soil tends to stay right around 32°F. This happens because of tiny pockets of air caught within the 6-sided crystals. This air insulates whatever is covered. Of course, too much snow on tree branches can lead to breakage. You made need to prune or protect trees if you get a lot of the white stuff. If temperatures happen to rise, well, plants get irrigated. Another surprising benefit is that nitrogen and sulfur in the atmosphere are captured as each crystal falls. When it melts, those important plant nutrients are released into the soil. If snow falls on your garden this winter, take a cue from Nature and grab yourself a nice fuzzy blanket, a hot beverage, and start planning your spring garden!
Jill Willard
12/24/2020 09:05:16 am
Enjoyed this article. Also great to hear that the publishing of your book is moving.
Kate Russell
12/31/2020 07:05:46 am
Thank you, Jill.
Keith (Chuck)
12/24/2020 11:25:16 am
Such a great article! Kudos too on getting your book published!
Kate Russell
12/31/2020 07:06:57 am
Thank you, Chuck! Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|



 RSS Feed
RSS Feed