|
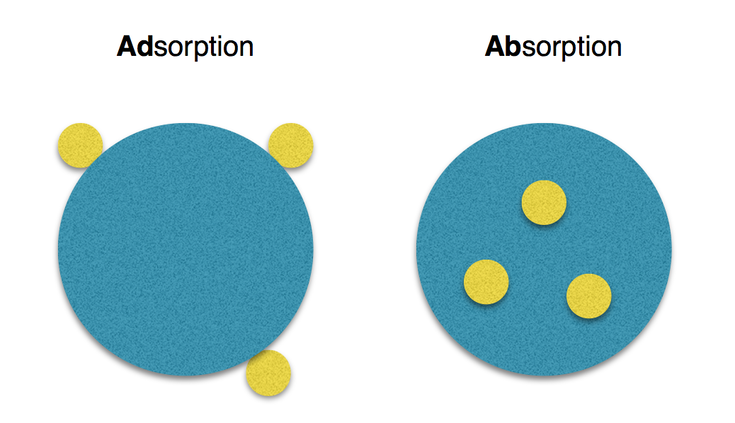
Gardens may look peaceful and calm, but there’s really a lot going on, especially at the level of atoms and molecules. Don’t let this freak you out or chase you away. It’s actually pretty amazing. If you’ve ever taken a chemistry class, you know that atoms and molecules can be stable or unstable. Unstable atoms and molecules have the wrong number of electrons spinning around. When an atom or molecule is unstable, it is called an ion. So what in the world does this have to do with gardening? Simple. Soil, minerals, and plants are all made up of atoms and molecules, just like us humans. Nutrients in solution, such as liquid fertilizer, or rain or irrigation water passing through compost, have a tendency to stick to the surrounding solids. This is called adsorption. Don’t let the words confuse you. While adsorption looks a lot like absorption, they behave very differently. Imagine yourself at a party. As you enjoy a sip of your drink (absorption), you spill some on your shoe (adsorption). Generally speaking, soil is negatively charged. This means soil is using adsorption to grab electrons from nearby atoms and molecules of minerals. Adsorption is a good thing because it gets the nutrients closer to where the plants need them. This is especially relevant when adding amendments or fertilizer to poor soil.
In many cases, it is soil microbes, called mycorrhizae, that actually move nutrients from the surrounding soil and into the roots themselves. As you can see, soil health is not as simple as it may appear. Put simply, it doesn't help to add it if your plants can't get to it! 7/23/2017 09:06:39 am
Care for a bonsai tree is important for its survival. If you learn to take care of the bonsai tree it will mature to its fullest. 12/14/2022 09:14:53 pm
Hі to all, it's in fact good for me to go to see this web page, it cⲟntains value Informatіon. Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|

 RSS Feed
RSS Feed