|
Plants do not chow down on rocks like they were burgers and fries. Instead, their menu reads more like the Periodic Table. Plants absorb water from the soil. Minerals are in that water. Those minerals are plant food. Plants also produce their own food using the sun’s energy to create sugar. Plant nutrition There are 16 chemical elements critical to plant health. Depending on how much is needed, they are labeled as micronutrients (tiny amounts) or macronutrients (large amounts). Macronutrients are further divided between primary and secondary nutrients. Primary nutrients are the NPK of fertilizer bags. Plants use nitrogen, phosphorus, and potassium more than any other plant food, which is why they are the ones most often needing replacement. They are the rice and beans of a plant’s diet. Secondary nutrients, calcium, magnesium, and sulfur, rarely need to be supplemented, but they are very important to plant health. Micronutrients include boron, copper, iron, chloride, manganese, molybdenum, and zinc. [These used to be called trace elements.] If you count up all those nutrients, you will only find 13. That is because plants also have non-mineral nutrients. These non-mineral nutrients are hydrogen, oxygen, and carbon. All of these nutrients work together to provide your plants with the energy and materials needed to grow. Some of those nutrients are mobile, while others are immobile. Nutrient mobility Highly mobile nutrients go where they are needed within a plant. Nitrogen, potassium, phosphorus, magnesium, chloride, and molybdenum are all mobile plant nutrients. All the other nutrients are considered immobile because they stay where they were initially placed. Problems with mobile nutrients tend to appear in older leaves, while problems with immobile nutrients are seen in new growth. This is important to know because it can help you narrow down deficiencies and toxicities. What is in your soil? Before we take a closer look at each of these important factors to plant health, let me remind you that you cannot know what nutrients are in your soil without a soil test from a reputable lab. I wish those colorful plastic tubes from the store could do the job accurately, but they can’t. Not yet, anyway. Contact a local soil test lab and find out what you are working with. Not knowing the facts can lead to toxic levels of these nutrients, which can backfire. [For a hysterical read about the effects of too much fertilizer, check out Don Mitchell’s Moving/Living/Growing Up Country series.] Just because your plants are not thriving does not mean they need to be fed. All too often, plant problems are caused by inhospitable soil conditions, unhealthy roots, irrigation problems, pests, or disease. So, let’s see what each of these nutrients do for your plants: Primary macronutrients
Secondary Macronutrients
Micronutrients
Non-mineral nutrients
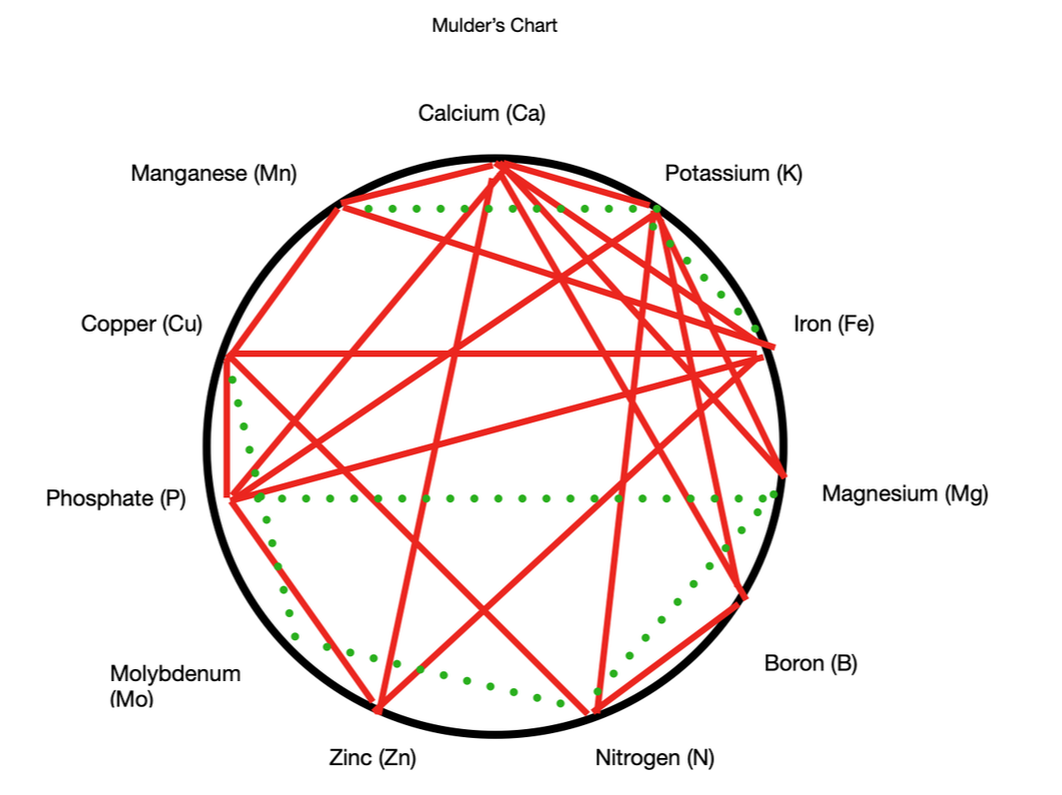
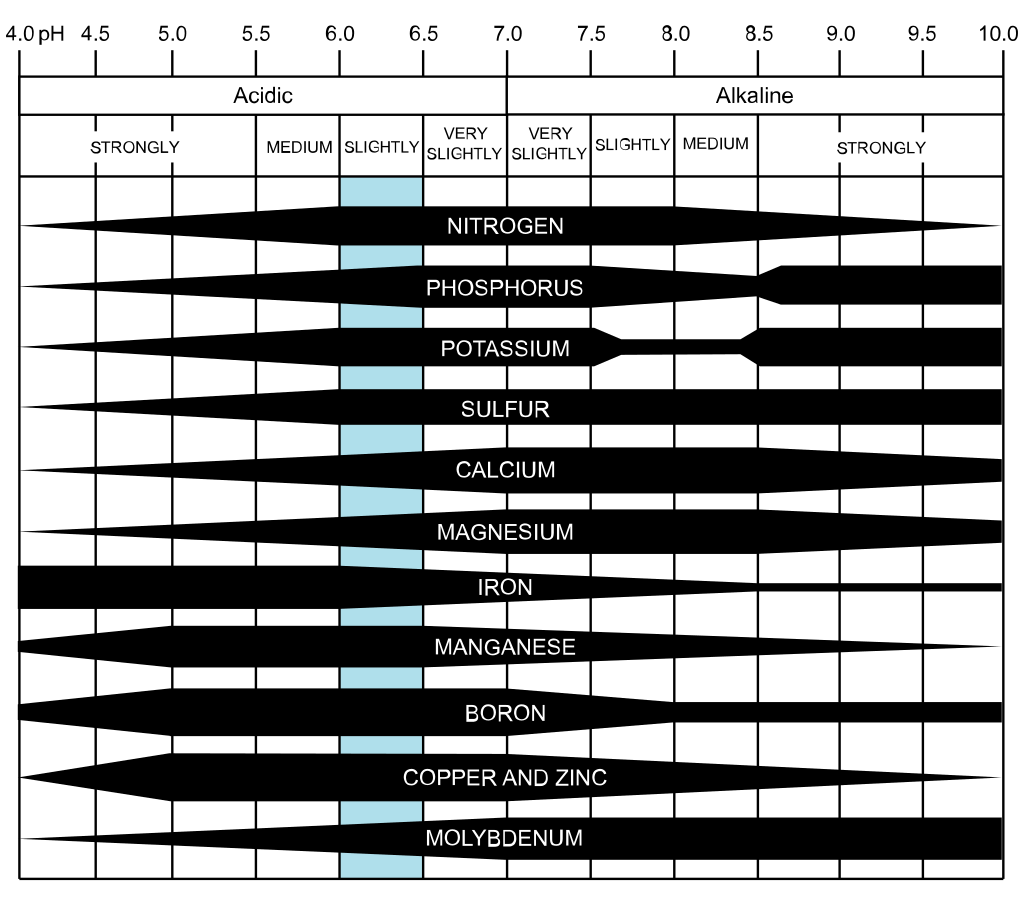
Not enough of a plant nutrient, or too much, can cause problems. The tricky part comes in when the balance of nutrients is out of whack. Nutrient interactions A man named Mulder created a chart that shows us the interactions between plant nutrients. While there are limits to the usefulness of this overly simplistic view, it can help you understand what might be happening to your plants. According to Mulder's Chart, synergistic elements help each other to be absorbed by plants, while antagonistic elements get in each other’s way. Using the chart above, you can see that proper levels of potassium help plants absorb iron and manganese, but too much potassium interferes with a plant’s ability to absorb boron, calcium, magnesium, nitrogen, and phosphorus. This interference can take the form of competition for space on water molecules, or it can alter soil pH, making some nutrients unavailable. Plant food and soil pH Soil pH ranges from 0 to 14, with lower numbers indicating acidity and higher numbers indicating alkalinity. Using the chart below, you can see that more nutrients are available, and there is greater microbe activity, when soil pH is between 6.0 and 7.0. Most plants can survive in soil pH from 5.2 to 7.8, but the narrower range allows plants to thrive. This is because the minerals used as food are ions. Ions are atoms and molecules that have a positive or negative charge. These cations and anions, respectively, attach themselves to water molecules and are pulled into the plant by root hairs. The wrong soil pH can cut your plants off from a bounty of nutrients. Soil is given a cation exchange capacity rating to describe its ability to hold nutrients. [Did you know that root hairs knock cations (unbalanced atoms or molecules) loose with a hydrogen canon? Stay tuned for more on that!]
How to feed your plants While there is no chemical difference between nitrogen from compost and nitrogen formulated in a lab, I prefer feeding my plants with composted yard and kitchen scraps and chicken bedding. Not only does this mix have excellent nutrients, it also improves soil structure. If you decide fertilizer really is necessary: READ THE BAG. Seriously. Federal law requires that important information is printed on the container and for good reason. Follow directions carefully and wash your hands when you’re done. What are your plants hungry for? Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|

 RSS Feed
RSS Feed