|
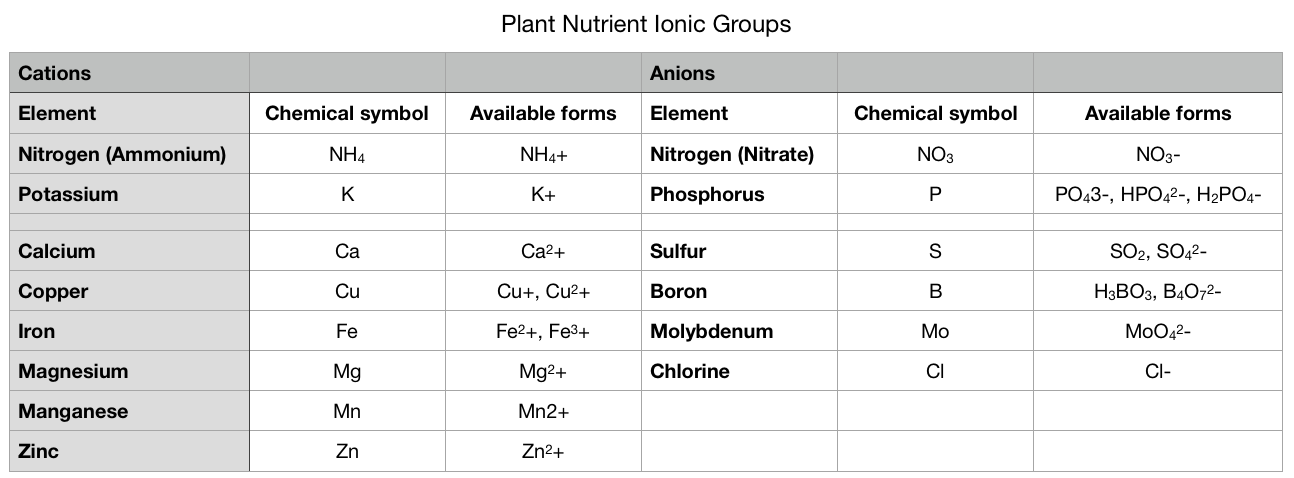
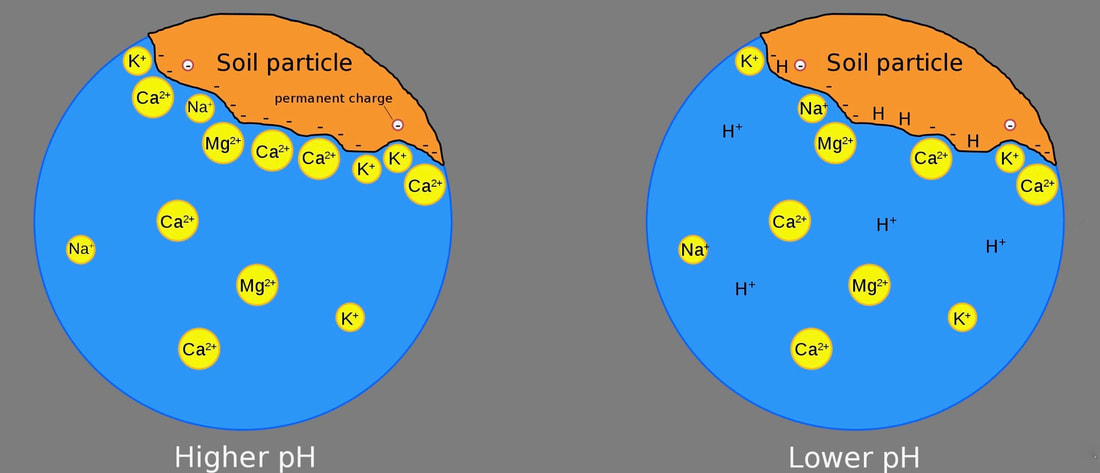
Cation exchange capacity (CEC) is a measure of soil fertility. The chemistry and science behind how and why a soil’s cation exchange capacity works the way is does is fascinating (and a little too complex for this venue). To put it simply, cation exchange capacity (CEC) is a measurement of how many positively charged minerals can be held by the surface of a soil particle. To learn how this affects the plants in your garden, we will need to touch up on some basic chemistry. Don’t panic - you can do this! Basic chemistry Everything is made up of atoms and molecules that are either positively charged (cations), negatively charged (anions), or neutral. Organic materials and clay tend to be negatively charged. This means these anions attract and hold cations, such as potassium, calcium, and iron, which are important plant nutrients. Many anions, such as phosphorus, sulfur, and boron are held in the water that is found in the spaces between soil particles. Soils with high CEC ratings generally hold onto more water, as well as nutrients. Below, you can see the electrical charge for each plant nutrient. Soil science Plant food exists as atoms and molecules of minerals. Minerals make up 45 to 49% of a soil sample. These mineral particles come in a range of sizes, with sand being the largest and clay being the smallest. Here in San Jose, California, we have clay. Chemically, sand is relatively unreactive and neutral. The spaces between particles (macropores and micropores) tend to be large. These spaces, and the lack of an electrical charge, make it more likely that plant nutrients will be leached out, or washed away. This is why sand has a low CEC rating. Clay, on the other hand, is made up of many negatively charged secondary minerals that love to attract and hold cations. Also, the smaller particle size of clay means that it has 100,000 times more surface area than sand, in the same size sample, so there are plenty of places for attachments to occur. Soil test results When I had my soil tested, it came back with a CEC rating of 20.6, but what does that mean? A CEC rating of 20.6 is considered relatively high. One way to look at a CEC rating is to think of it as a power strip - just how many cords can be plugged in? CEC is measured using mEq/100g. We won’t get into it, but it basically means how many parts of something there are in a certain volume of soil. Different soil types have very different CEC ratings: My soil’s CEC rating of 20.6 means it can hold onto a significant amount of plant nutrients. Another, related figure found on a soil test is called base saturation. Base saturation is the percentage of available connections being used. You will normally see separate figures of base saturation for calcium, magnesium, and potassium. [You can think of them as different sized electrical plugs.] Below, you can see my base saturation results with “values found” (left) and “optimal ranges” (right). CEC and pH Soil pH also plays a role in a soil’s ability to hold onto plant nutrients. This is because pH is a function of rogue hydrogen cations (H+) floating around in the soil. Soils with a higher, more alkaline pH, tend to have a higher CEC rating. Of course, too much of a good thing turns out to be a bad thing. If the soil becomes too alkaline, nothing can grow in it! This is true in other ways, as well. Too much ammonium (NH4+) in the soil can interfere with the uptake of potassium (K+), calcium (Ca2+), and magnesium (Mg2+). This is why soil tests are so important. Armed with the information they provide, you can look at fertilizer labels with a more informed idea of what your soil actually needs. Acidifying our local clay is one way to make more nutrients available to your plants. The opposite is true in areas with acidic soil. There is a happy medium, but soils with a higher CEC rating are more difficult to alter, when it comes to pH. Bottom line, cation exchange capacity is a measure of your soil’s negative charge which, in turn, tells you just how many nutrients it can hold at any one time.
Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|


 RSS Feed
RSS Feed