|
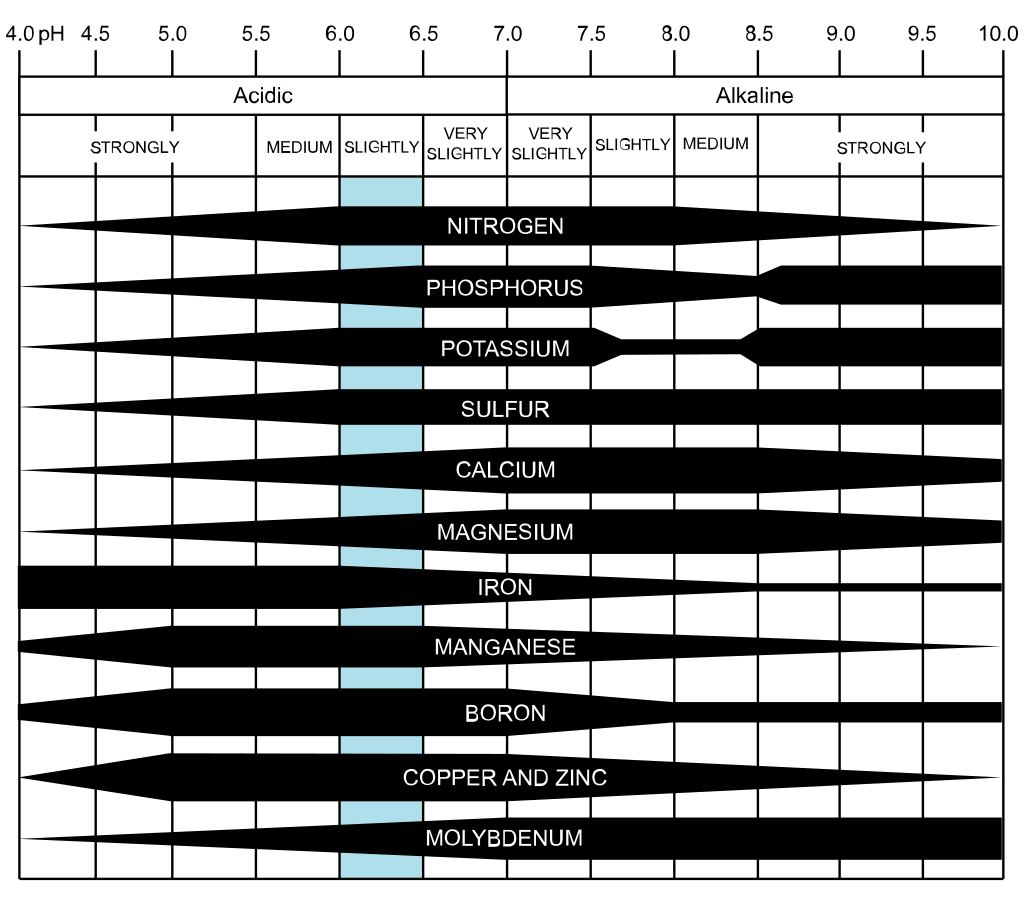
Micronutrients are elements that plants only need in small (micro) doses. These used to be called trace elements, but the American Society of Agronomy and the Soil Science Society of America are urging us to use the term micronutrient instead. Whatever you call them, your plants need them to grow and produce. Plant nutrition Like us, all plants need water, oxygen, and carbon to live and thrive. They also need the Big Three: nitrogen, phosphorus, and potassium. Beyond that, to function properly, plants need secondary nutrients and micronutrients to stay healthy. All together, these are called essential nutrients. But, before we learn about the various players in the world of plant nutrition, you need to understand that nutrient uptake is a delicate, intricate dance that occurs between root hairs, soil minerals, moisture, soil pH, and some mind-blowing processes, all of which are occurring at the molecular level! Did you know that root hairs knock positively changed ions loose with a hydrogen canon? Stay tuned for more on that! What’s important to keep in mind is that too much of a good thing can be a bad thing. Essential nutrients can reach toxic levels, causing just as much damage as not having enough. To make matters even more confusing, the symptoms of one nutrient’s toxicity make look exactly like a different nutrient’s deficiency. And none of this occurs in isolation. A deficiency in one nutrient can domino into the deficiency of several other nutrients. Also, some plants can block the entry of some nutrients, preventing toxicity, while others cannot. If that weren’t confusing enough, some nutrient deficiencies and toxicities can look exactly like herbicide overspray damage, pest feeding, or a disease. Fear not! We will sort this out as best we can. Read on! Plant nutrients If you don’t know what your plants need, you can’t give it to them. A basic understanding of plant nutrition can help you help your plants. For a quick refresher of the three macronutrients: Nitrogen (N) - used in photosynthesis for rapid leaf growth Phosphorus (P) - used in photosynthesis for flower, fruit and root growth Potassium (K) - used to fight disease and improve fruit quality Secondary nutrients also play vital roles in plant health: Calcium (Ca) - used for cell wall structure and to move other nutrients around Magnesium (Mg) - essential for photosynthesis and it activates plant enzymes Sulfur (S) - used to create amino acids for root and seed production For plants, secondary nutrients and micronutrients work much the way vitamins do for us: you probably won’t die without them, but your teeth may fall out and you certainly won’t be at your best. Nutrient mobility As you learn about plant nutrients, you need to know that some of them are free to move around within a plant and others are not. Highly mobile nutrients, such as nitrogen and potassium, go where they are needed. This means that deficiencies are seen in older growth, as the nutrients are pulled away to provide for new growth. The opposite is true for nutrients that do not readily move around. Once they are absorbed, they tend to stay where they are. This means that deficiencies are usually seen in young leaves and new buds. Below, you will find a brief description of each plant micronutrient. This will help you to understand how these nutrients are used, and what the plants look like if they are missing a nutrient (or have too much). Aluminum (Al) Aluminum acidifies the soil by removing hydroxide ions out of water, which leaves acidic hydrogen ions behind. Aluminum is not exactly a plant nutrient, though it is believed to act as a fungicide for some root rots. The reason for its inclusion is that it can be absorbed by plants to the point of toxicity. Aluminum toxicity causes slowed root growth. At the same time, aluminum is frequently applied to tea crops because of the way it prevents copper, manganese, and phosphorus toxicity. Boron (B) Boron is used to make cell walls. It also helps plants use and regulate other nutrients by facilitating the production of sugar and carbohydrates. Boron helps plants reproduce. This means plants use boron to flower, fruit, and go to seed. It is also used in pollen generation and cell division. Boron is only available to plants when the soil pH is between 5.0 and 7.5. [My last soil test came back with a pH of 7.7, which is not uncommon in the Bay Area.] Boron is generally created by decomposing organic matter that is deposited on the soil surface. Unfortunately for many of our summer plants, boron cannot be absorbed once the soil is dry. Boron is an immobile nutrient that works in tandem with calcium, so a deficiency of one can lead to the unavailability of the other. Boron deficiency appears as stunted, crinkled, or otherwise distorted fruit or buds (meristem tissue), dark rings on leaf petioles (those tiny stems that attach leaves to twigs), roots that are shorter, thicker, and highly branched, or upper leaves that turn reddish yellow. Too much boron in the soil makes leaves look scorched, with browned areas on leaf tips and edges. Chlorine (Cl) Chlorine aids plant metabolism during photosynthesis. It is necessary for osmosis and fluid balance within plants. It is a mobile micronutrient. Too much chlorine in the soil, common in areas with hard water, can interfere with a plant’s ability to absorb nitrogen. [San Jose tap water ranges in pH from 7.0 to 8.7.] Cobalt (Co) Cobalt is not needed by all plants. It is only used by legumes in nitrogen fixing. Copper (Cu) Copper is used to make reproductive enzymes - that means flowers, fruits, and seeds. Copper also helps plant roots eat and breathe, and it metabolizes proteins. Since copper is not mobile inside the plant, deficiencies are usually seen near the top or in new growth, rather than established leaves and stems. Copper deficiencies can cause leaf rolling and curling. Iron (Fe) Iron is essential in the production of chlorophyll and moving electrons around within the plant. Iron is also used in enzyme functions that help your plants absorb many other nutrients. This means your soil, like mine, can contain plenty of everything else, but insufficient iron makes it difficult for plants to get at all that food. Iron deficiencies usually appear as chlorosis (yellowing) and necrosis (dying) between young leaf veins, especially at the top of the plant. Iron is not readily mobile. If your soil is alkaline or deficient in copper, it can make an iron deficiency even worse. Manganese (Mn) Manganese is used to make chlorophyll and activates plant enzymes that breakdown carbohydrates and nitrogen into usable bits. It is also used as an antioxidant. Manganese is not mobile, so symptoms of deficiency are usually seen near the top of the plant. Yellowing between the veins of young leaves, with tan flecks, while the areas next to veins stays dark green, is the first symptom of a manganese deficiency. Deficiencies are more likely in alkaline soil. Too much calcium can cause a manganese deficiency, which can, in some cases, be counteracted by adding more nitrogen. Molybdenum (Mo) Molybdenum helps plants use nitrogen by working with certain enzymes. If molybdenum is deficient, or if any of those enzymes become sluggish, overall plant growth will slow significantly. Molybdenum is partially mobile. Nickel Nickel is used to activate certain enzymes. Insufficient nickel causes a condition called mouse ear, in which stems are shorter than normal and leaves are smaller and more rounded. Insufficient nickel allows urea to collect within plants, causing lesions. Selenium Selenium isn’t exactly an essential nutrient, but plants grown on selenium-depleted soils end up being less nutritious for us. Selenium is believed to stimulate plant growth and to counteract stress, pests, and disease. [In the human body, selenium makes antioxidant enzymes that prevent cell damage.] Silicon Silicon is found everywhere, so we generally do not think of it as an essential nutrient. That being said, silicon is used by plants to build cell walls and to improve plant health and productivity. Silicon is believed to help plants counteract drought and frost damage. Sodium Plants, like us, can suffer or even die from too much sodium, though, for them, it’s not from heart failure. Sodium can replace other critical nutrients within a plant, potassium and nitrogen, in particular. At the same time, low levels of sodium are needed by plants to stimulate growth, maintain a water balance, and improve fruit flavor. Vanadium Vanadium is not used by all plants. When it is used, plants are using it as a substitute for molybdenum. Zinc (Zn) Zinc helps plants breakdown carbohydrates, and it regulates sugar consumption. Zinc is also used to activate enzymes. It is not mobile. Zinc deficiencies show up as yellowing between young leaf veins and overall bleaching that does not reach leaf edges or midribs. This bleaching can also take the form of narrow yellow or white stripes between the veins of upper leaves. Zinc deficient leaves may also roll or curl, and leaves may be smaller than normal. Zinc deficiencies are common in areas with alkaline soil and/or insufficient organic matter. (So keep composting!) Don’t be surprised if you are feeling a bit overwhelmed with all that information. We all do. And the more we learn, the more amazing all these interactions turn out to be. It’s really pretty spectacular. And, all you need to take away from this is that monitoring your plants for changes can help you better understand what they need. As you work in your garden, be sure to ask yourself these questions:
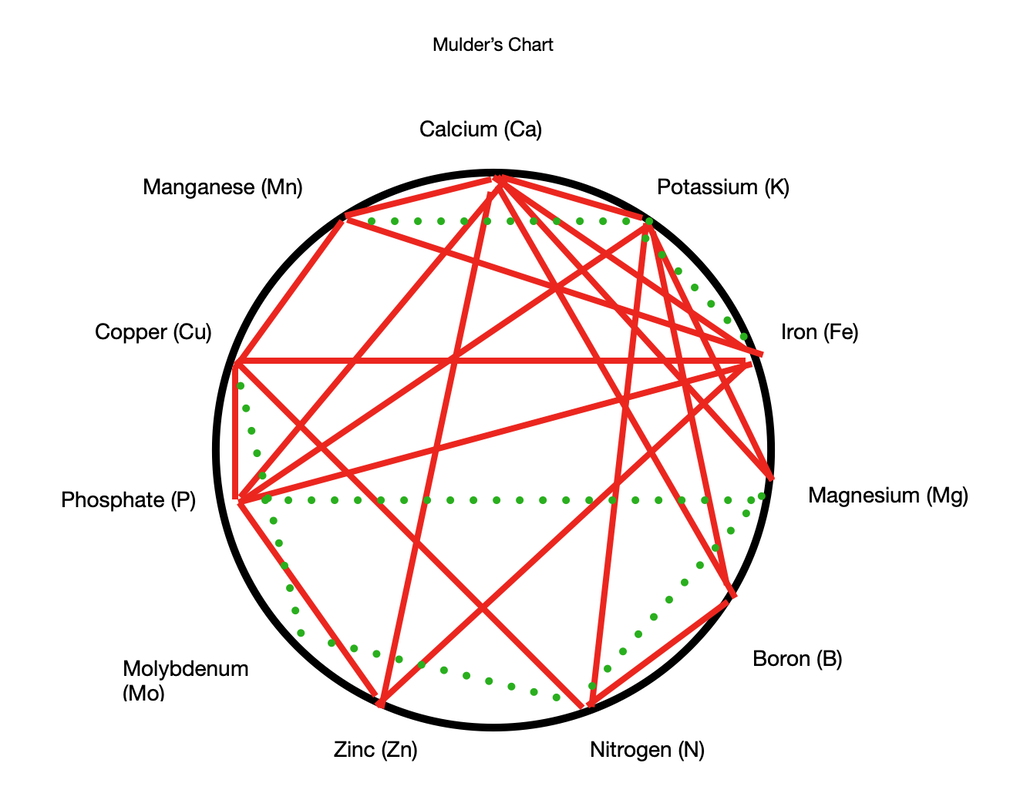
The answers to these questions, combined with your own observations, will help you to identify problems. Finally, I discovered this interesting perspective on plant nutrient interactions: Mulder’s Chart
Back in 1953, a man named Mulder created a chart that shows how he believed different nutrients interact. The relationships shown in the graph are either antagonistic (red lines) or enhancing (green lines). It takes a little getting used to, and we will discuss it further, but give it a ponder. To get you started, take a look at the lines that point to potash (potassium). You can see green lines connecting it to iron and manganese. That means the presence of potassium, iron and manganese facilitates the uptake of all three, simply because of their chemical makeup. At the same time, red lines can be seen connecting potassium to boron, calcium, magnesium, nitrogen, and phosphorus. This means that potassium competes with these elements on the root hairs. It also alters soil chemistry, making it so alkaline that iron and boron cannot be absorbed. Of course, there are dozens of other variables at play, so this is an oversimplified explanation of what's happening in your soil, but it might get you to take a closer look at your soil test results! What are your plants trying to tell you about the soil they live in? Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|




 RSS Feed
RSS Feed