|
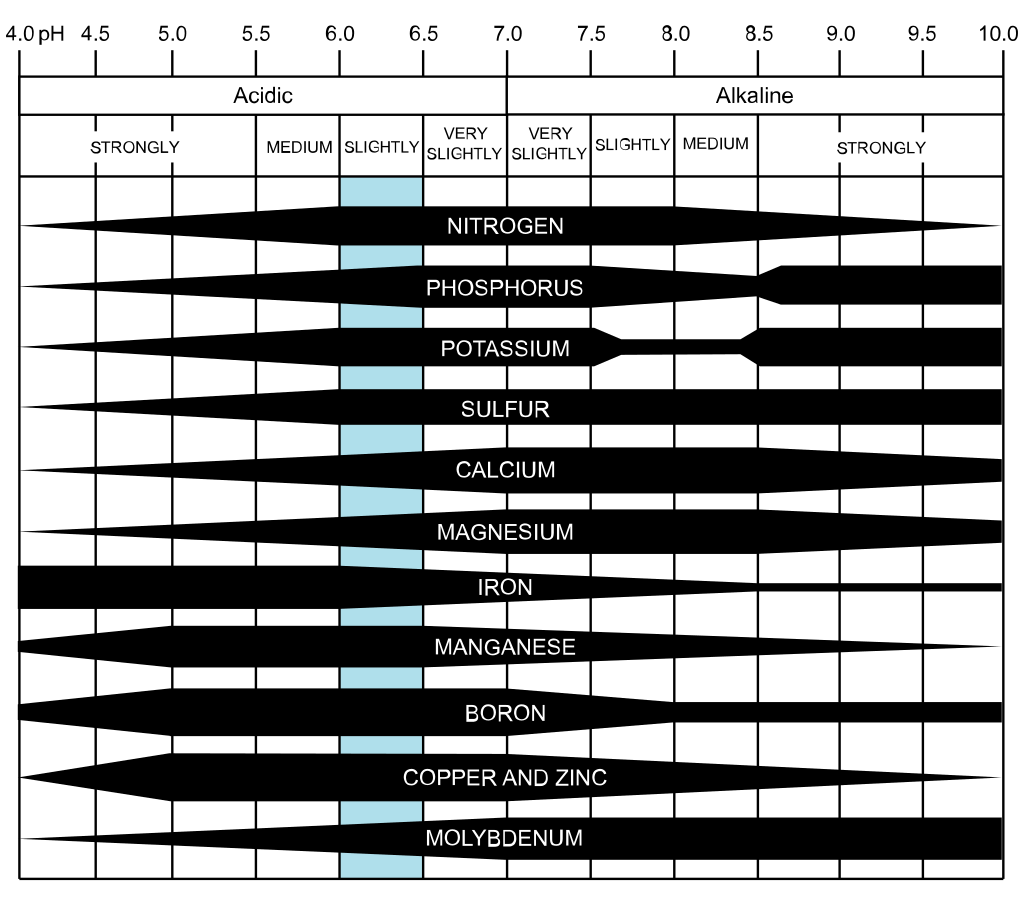
You may see the word chelated [ˈkēlāted] on bags of fertilizer, but what does that mean, and how can it help (or harm) your garden? Advertisers make many claims about chelated fertilizer. They say it will help your plants thrive and reduce chlorosis by making more soil nutrients available to plants. In some cases, chelates hold onto ions that pathogens need to growth, thereby reducing their population. These claims are all accurate, mostly. What they don’t tell you is that the effectiveness of chelated products depends entirely on soil pH and that many chelated products are rather toxic to both the environment and to soil microorganisms. Before adding chelated fertilizers to your food crops, let’s find out what’s really going on, shall we? The chemistry of chelation Chelates (or ligands) are organic molecules (chemically speaking) that attach themselves to metal minerals found in the soil. The word ‘chelate’ comes from the Greek word for lobster’s claw (chelé). This word describes the claw-like mechanism that surrounds a metal nutrient and ‘complexes’ the mineral. This prevents the mineral from being oxidized or precipitated. Once a mineral is complexed, it is held until a root hair is reached. Then the mineral is released and absorbed by the root hair, and the chelate goes in search of another mineral. [Try wrapping your brain all of those actions taking place around the roots of all your plants… it’s mind boggling!] Metal minerals as plant food Metal micronutrients, such as copper (Cu), iron (Fe), magnesium (Mg), manganese (Mn), and zinc (Zn), are important food for your plants. But plants can only use these metals when they are in the form of water soluble ions. Very often, these molecules attach themselves to oxygen molecules and become oxidized, or hydroxide ions (OH-) to be precipitated. This makes them unavailable to plants. Both oxygen and hydroxide ions are abundant in the soil. When metal nutrient molecules become chelated, they can be absorbed and used by plants. In fact, plants produce some of their own chelates. This must mean chelation is a good thing, right? Well, not always. The power of pH If your soil has a pH greater than 6.5, chelated fertilizer can provide plants with the nutrients they need in low quality soil without risking eutrophication. [Eutrophication refers to the condition of too many nutrients ending up in bodies of water due to urban drool and runoff, which leads to dense plant growth and animal death due to lack of oxygen. It’s not pretty.] Did you know that different minerals become unavailable at different soil pH levels? No simple answers
Here is where I have my biggest concern about chelated products. All too often, people see plants not growing well, so they add fertilizer. The plants don’t improve fast enough, so more fertilizer is added. This cycle can continue indefinitely because the lack of all those nutrients isn’t the problem. Very often soil structure and soil health are the problem. It could be a lack of soil microorganisms, or the incorrect choice of plants for a certain microclimate. Frequently, the lack of a single nutrient, usually iron on the West Coast and calcium on the East Coast, can make all the other nutrients unavailable. Without soil test results from a reputable, local lab, there is no way of knowing what your soil needs. Before adding chelated fertilizers, you need to recognize that there’s much more to this situation than the Quick Fix claims. According to a report published by the University of Florida Extension Office, you need to take into account soil pH, your soil’s bicarbonate content, and the specific plant species when deciding whether or not to use chelated products. Bicarbonate levels? What does that mean? Bicarbonates are ions that precipitate (attach to) calcium, leaving salt behind. If your soil or irrigation water have a pH of 7.5 or higher, you can safely assume that the bicarbonate levels are higher than is good for your plants. Are you sure you know what you’re doing? It’s not nearly as simple as advertisers make it out to be. [Is it ever?] And where do those chelates come from? Chelate sources Chelated products normally include ingredients such as DTPA, EDDHA, and EDTA. Since iron is the most commonly chelated mineral, we will look at the effects of those ingredients on soil health and iron absorption:
Not all bad There are times when the use of chelated fertilizers make sense, if used judiciously. For example, chelated iron helps you grow acid-loving plants, such as blueberries, in alkaline soil. Start by selecting the best form of chelate for your soil pH. You can avoid soil-based problems by using foliar (leaf) applications of chelated products, once you are certain of the need for them. You can also increase the number of naturally occurring chelates (and improve soil health) by adding more organic material in your soil. This is done by top dressing the soil with aged compost or mulching with clean wood chips. Just remember, quick fixes that sound too good to be true are usually far more complex and risky than they are made out to be. Comments are closed.
|
Welcome!You can grow a surprising amount of food in your own yard. Ask me how! To help The Daily Garden grow, you may see affiliate ads sprouting up in various places.
You can also get my book, Stop Wasting Your Yard! Index
All
Archives
July 2024
|

 RSS Feed
RSS Feed